AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Asymmetric carbon atom8/26/2023 An asymmetric carbon is often a chiral stereogenic center, since interchanging any two substituent groups converts one enantiomer to the other. Stereogenic elements may be chiral or achiral. A stereogenic element is a center, axis or plane that is a focus of stereoisomerism, such that an interchange of two groups attached to this feature leads to a stereoisomer. This aspect of stereoisomerism will be treated later.Ī useful first step in examining structural formulas to determine whether stereoisomers may exist is to identify all stereogenic elements. When more than one chiral center is present in a molecular structure, care must be taken to analyze their relationship before concluding that a specific molecular configuration is chiral or achiral. Most of the chiral centers we shall discuss are asymmetric carbon atoms, but it should be recognized that other tetrahedral or pyramidal atoms may become chiral centers if appropriately substituted. The presence of a single asymmetrically substituted carbon atom in a molecule is sufficient to render the whole configuration chiral, and modern terminology refers to such groupings as chiral centers. Consequently, we find that these compounds exist as pairs of enantiomers. The structural formulas of lactic acid and carvone are drawn on the right with the asymmetric carbon colored red. In the general figure below, A and B are nonsuperposable mirror images of one another, and thus are a pair of enantiomers. 
This type of configurational stereoisomerism is termed enantiomorphism, and the non-identical, mirror-image pair of stereoisomers that result are called enantiomers. The configuration of such a molecular unit is chiral, and the structure may exist in either a right-handed configuration or a left-handed configuration (one the mirror image of the other). A regular tetrahedron several planes of symmetry and is achiral.Ī carbon atom that is bonded to four different atoms or groups loses all symmetry, and is often referred to as an asymmetric carbon. If all four substituent groups are the same, as in methane or tetrachloromethane, the configuration is that of a highly symmetric "regular tetrahedron". 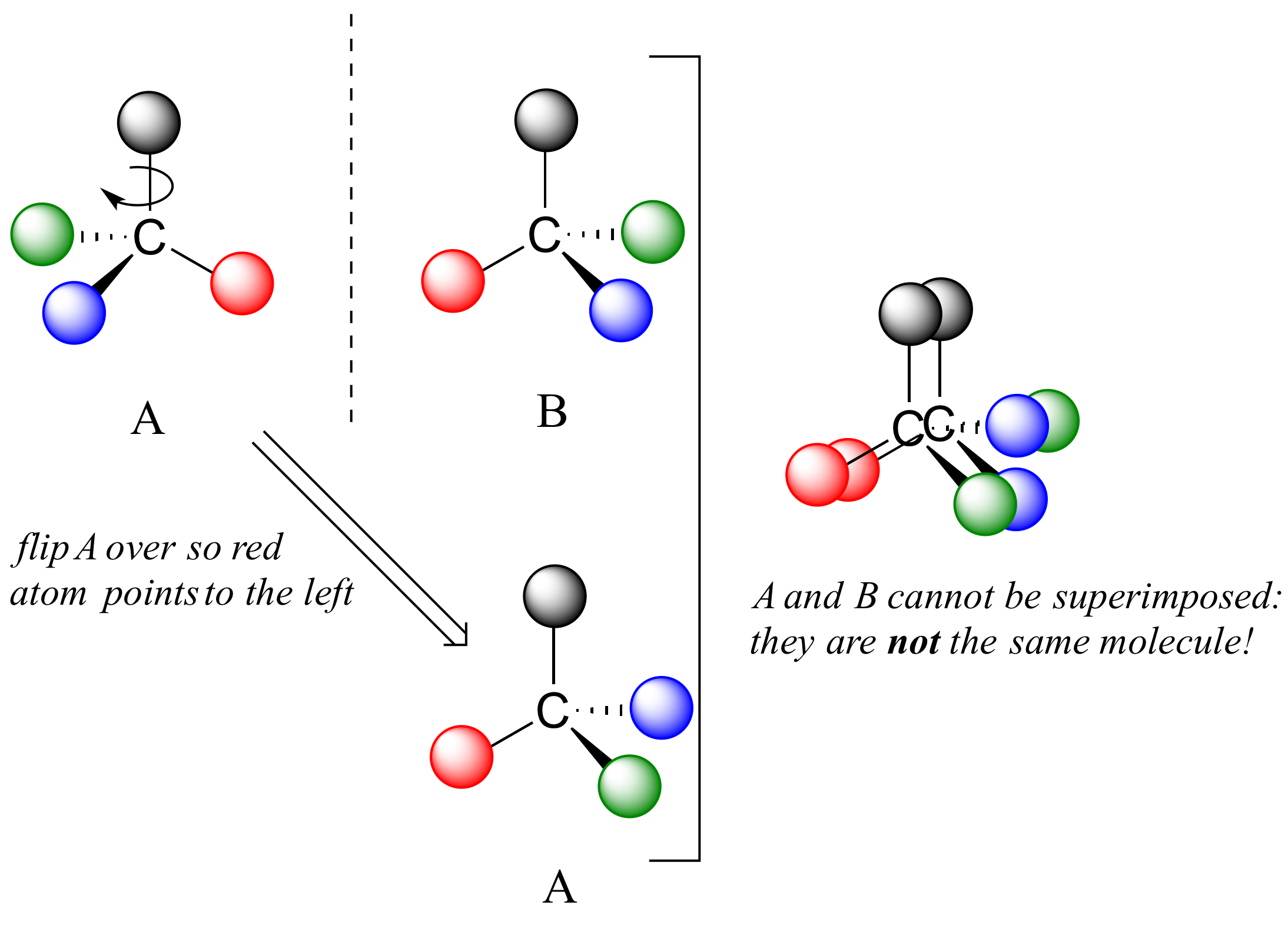
Tetravalent carbons have a tetrahedral configuration. The results of mechanistic studies, including the isolation of a chiral heteronickelacycle, support that the enantioselectivity on the two contiguous carbon atoms of the γ-lactams is determined during the oxidative cyclization on nickel(0).\)Ī consideration of the chirality of molecular configurations explains the curious stereoisomerism observed for lactic acid, carvone and a multitude of other organic compounds. A key of the procedure presented here is a nickel(0)/chiral phosphoramidite-catalyzed asymmetric carbonylative cycloaddition between readily accessible ene-imines and carbon monoxide, which proceeded enantioselectively to furnish up to 90% ee (>99% ee after recrystallization). Moreover, we have achieved the first enantioselective synthesis of strigolactam derivative GR-24, a racemic variant of which is a potential seed germination stimulator and plant-growth regulator. 
Herein, we report a strategy to construct polycyclic γ-lactam derivatives that contain more than two contiguous stereogenic centers in an enantioselective as well as atom-economic manner. The development of straightforward and reliable synthetic routes to such chiral structural motifs in a stereocontrolled manner should thus be of importance. Γ-Lactam derivatives with multiple contiguous stereogenic carbon centers are ubiquitous in physiologically active compounds.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
